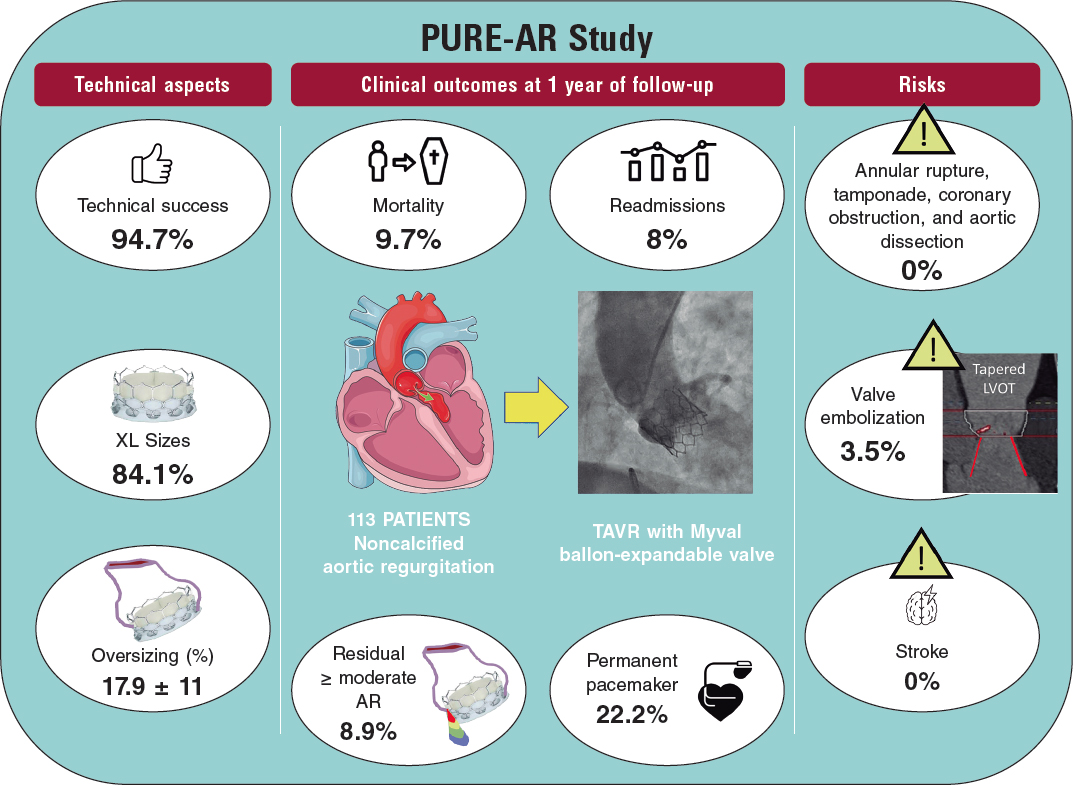
HOW WOULD I APPROACH IT?
Authors describe the case of a 51-year old patient with obstructive hypertrophic cardiomyopathy with unusual clinical presentation and progression. The patient had repeated episodes of sustained monomorphic ventricular tachycardia (VT) that required defibrillator implantation in secondary prevention and pharmacological treatment with amiodarone and beta-blockers.
Afterwards, the patient experienced an arrhythmic storm with appropriate multiple discharges despite the antiarrhythmic medication. The stellate ganglion block was indicated. Modulating the sympathetic nervous system can be effective to suppress the arrhythmic storm, but I think of it as the last option. The first therapeutic strategy here would have been the electrophysiological study and the substrate-based ablation of VT. The ablation of recurring VT has a high rate of success with obstructive hypertrophic cardiomyopathies with apical aneurysm and a clear anatomical substrate. However, when this is not the case and there are several fibrotic areas like this patient’s cardiovascular magnetic resonance (CMR) imaging showed, the utility of ablation is limited. According to the electrocardiogram and late gadolinium enhancement CMR imaging, the origin of VT could be septal. The authors describe that the intracardiac mapping showed septal macroreentrant whose ablation failed. The endocardium catheter ablation is expected to fail in cases like this when the focus is at deep intramyocardial location. If ablation through endocardial and epicardial accesses fails, the next step is trying alcohol septal ablation.
Back in 1989, Brugada et al.1 described for the first time the successful management of refractory VTs using intracoronary injections of alcohol in humans. From that moment on, isolated cases and series of patients2 have been published supporting the use of alcohol septal ablation as an effective alternative in these cases. However, recurrence is still high.2
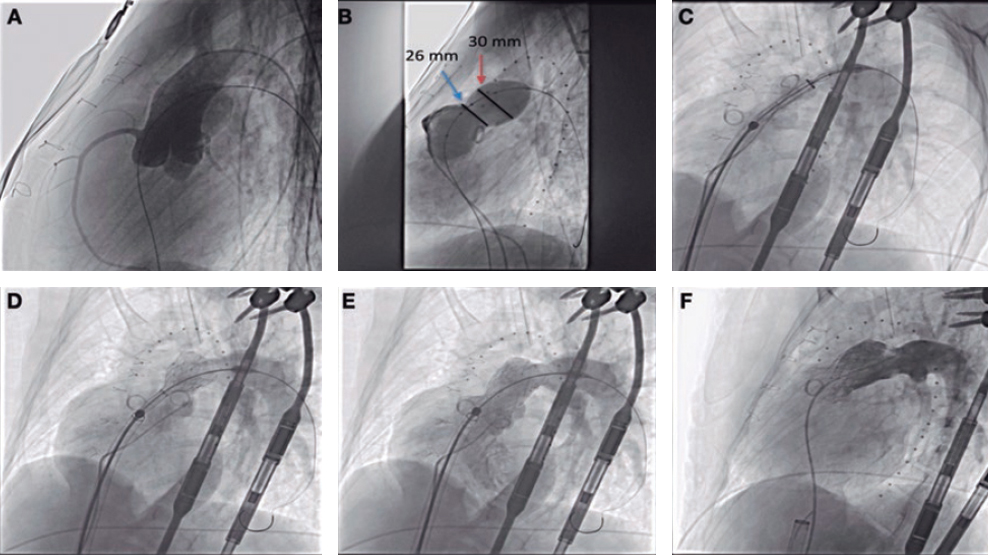
When available a CT scan with contrast can be used to study the origin and distribution of septal arteries. Combining the CT scan imaging and the CMR imaging showing the fibrotic area we can identify the septal branches that irrigate the arrhythmogenic focus to plan the alcohol septal ablation procedure.3 This procedure should be planned by the electrophysiology, hemodynamics, and cardiac imaging teams combined. Using the venous access, the electrophysiologist introduces the catheters for mapping purposes and stimulates the VT. The interventionist performs a routine alcohol septal ablation procedure through 2 arterial accesses (right femoral of 7-Fr for the guide catheter and right radial of 5-Fr for the pigtail catheter) plus the simultaneous registry of pressures. In this patient, the CMR imaging showed late enhancement in the mid portion of intraventricular septum, so it was not probably the first but the second septal branch that irrigated the arrhythmogenic focus (figure 3A, figure 3B of the case presentation). After the coronary angiography, a long 0.014 in guidewire is introduced in the target septal branch and a short and relatively large coaxial balloon compared to the septal branch (at least in a 1.2:1 ratio). When the balloon has been inflated at low atmospheres inside the septal branch and the guidewire has been removed, 1-2 mL of echocardiographic contrast are injected through the balloon lumen to discard collaterals. Using the transthoracic echocardiography (TTE) it is verified that the target focus of the septum opacifies (where the origin of the VT would be); in this case, the medial-basal portion of the septum. At this point, VT is induced. The ideal scenario would be that with the inflation of the balloon and the corresponding ischemia, the VT was suppressed, suggestive that alcohol septal ablation may be effective. If this is not the case, the administration of 2-3 mL of a cold physiological saline solution through the balloon can be effective too. Then using ultrasound guidance alcohol is gradually injected through the balloon in less than 1 mL per 1 cm of septal thickness. After the alcohol infusion, the balloon is kept inflated for 10 minutes and the lack of complications in the coronary artery tree is verified; we often seen that the septal branch treated has no flow. VT is re-induced again but if unsuccessful, the procedure is considered terminated. Not being able to induce VT is not a guarantee for future success. But if VT is actually induced, the procedure can be repeated using a different septal branch—the distal sub-branch of the first septal branch—to induce ischemia-necrosis in the edges of the area already treated.
While the procedure is being performed targeting the ablation of the focus of VT, the pressure gradient is being monitored. The procedure may also be useful to reduce the subaortic dynamic gradient, but we should remember that we need to wait for a full year before confirming that it has been effective. However, if the gradient does not fall in the cath. lab, we can use the procedure to test the first septal branch, inflate the balloon in its proximal sub-branch, for ultrasound-guided contrast infusion, to see if the basal septum opacifies in contact with the anterior mitral leaflet and to see what’s happening with the gradient. If the gradient does fall, we would have confirmation that such septal branch is causing the gradient, which is important for an eventual procedure if the patient’s functional class gets worse.
The most common complication is atrioventricular block that has a higher prevalence in alcohol septal ablation (ASA) procedures of VT compared to procedures to reduce the gradient: 35% vs 5% to 10%.2 In the absence of complications, the patient will stay 2 days in the coronary unit with a temporary pacemaker and another 5 at the hospital ward with telemetry.
REFERENCES
1. Brugada P, de Swart H, Smeets JL, Wellens HJ. Transcoronary chemical ablation of ventricular tachycardia. Circulation. 1989;79:475-482.
2. Tokuda M, Sobieszczyk P, Eisenhauer AC, Kojodjojo P, Inada K, Koplan BA. Transcoronary ethanol ablation for recurrent ventricular tachycardia after failed catheter ablation. An update. Circ Electrophysiol. 2011;4:889-896.
3. Roca-Luque I, Rivas-Gándara N, Francisco-Pascual J, Rodriguez-Sanchez J, Cuellar-Calabria H, Rodriguez-Palomares J. et al. Preprocedural imaging to guide transcoronary etanol ablation for refractory septal ventricular tachycardia. J Cardiovasc Electrophysiol 2019;30:448-456.
Corresponding author: Unidad de Cardiología Intervencionista, Planta 0 izquierda, Hospital Ramón y Cajal, Ctra. Colmenar Viejo km. 9,100, 28034 Madrid, Spain.
E-mail address: recalde@secardiologia.es (Á. Sánchez-Recalde).