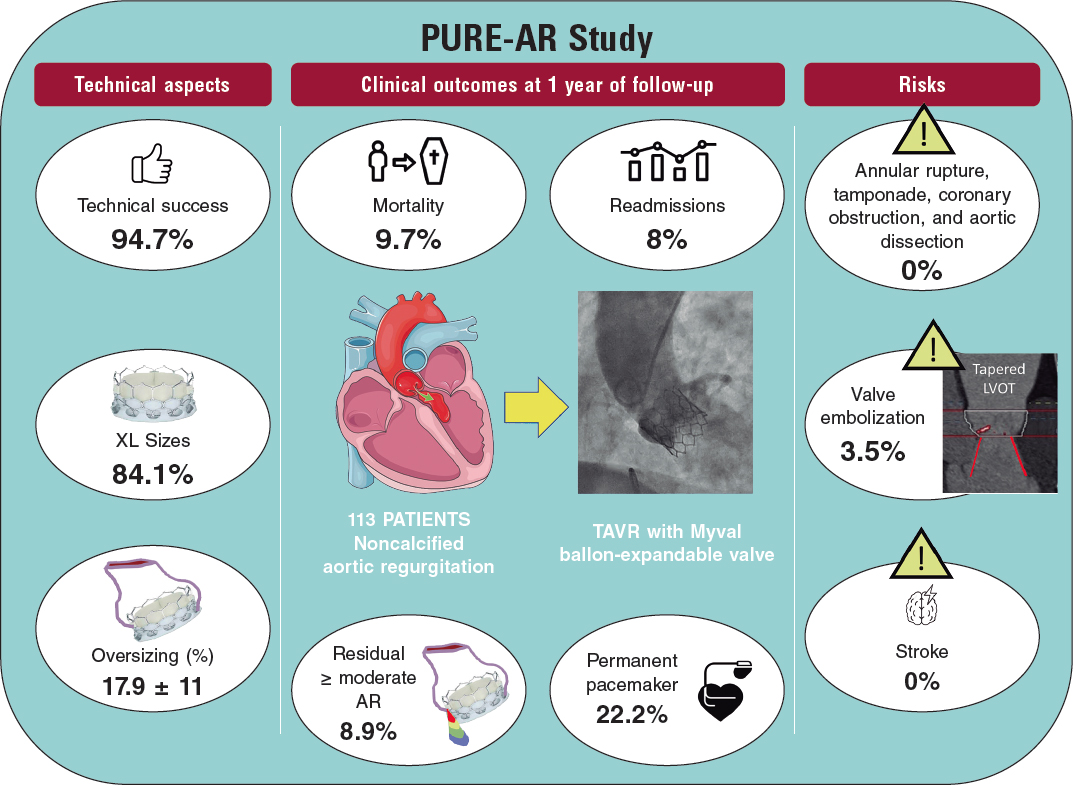
CASE RESOLUTION
The evolution of the patient was not favorable, brain death was declared 24 h after admission, and the patient was eventually declared dead. The microscopic analysis of the coronary aspirate revealed the presence of mesenchymal tissue with myxoid changes consistent with tumor embolism (figure 1) whereas the macroscopic piece confirmed the diagnosis of atrial myxoma (figure 2, arrow).
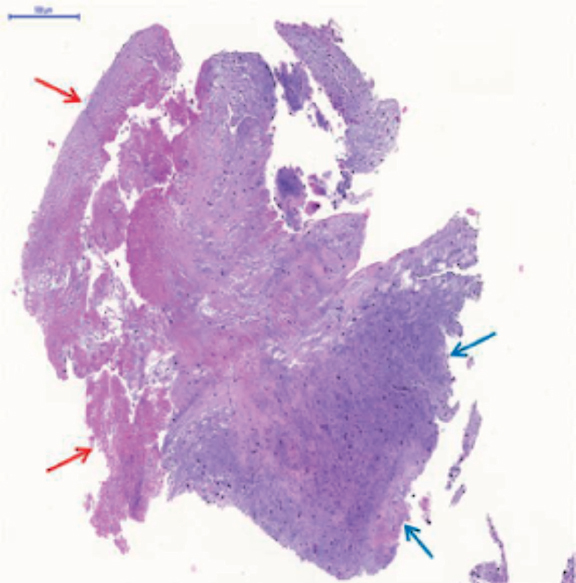
Figure 1. The red arrows point at the fibrinoid tissue. The blue arrows point at the mesenchymal tissue with myxoid changes.
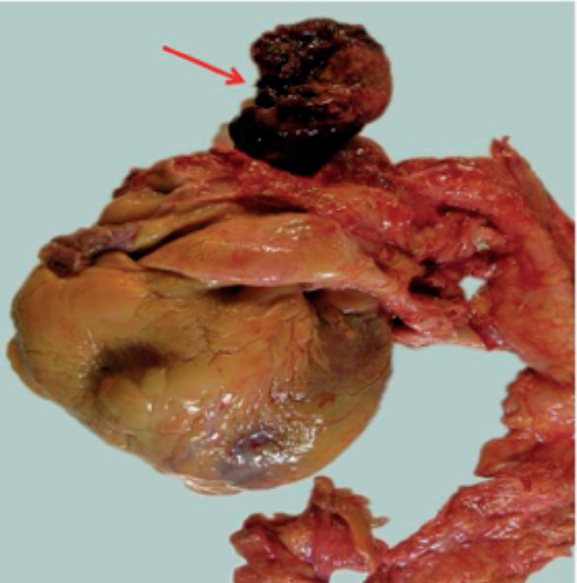
Figure 2. Atrial myxoma, macroscopic piece.
Although they are regarded as benign tumors, cardiac myxomas can lead to life-threatening events. Among their different ways of presentation, coronary embolism is one of the least common of all (0.06%). One possible explanation to this low incidence can be found in the anatomical-functional characteristics such as the existing straight angles between the aortic root and the coronary ostia, the protection of coronary arteries by the cusps of the aortic valve, and the coronary filling during ventricular diastole.
The shape of the tumors is one of the leading predictors of embolization, and papillary or villous myxomas are thought to have the highest potential to cause embolisms. According to the medical literature available, the right coronary artery is the most common location of embolization; however, there is an interesting percentage of normal coronary arteries on the angiography1 probably attributed to the fact that the myxomatous histology of the tumor favors greater fragmentation, distal spread, and spontaneous resolution. The clinical signs of tumor embolism go from silent events to ST-segment elevation acute myocardial infarctions (STEMI) complicated with cardiogenic shock, as it was our case, and the emergent therapy here is similar to the management of atherosclerotic coronary syndromes.
We should emphasize that the use of mechanical circulatory assist devices in the setting of STEMI-related cardiogenic shock is not recommended systematically (evidence IIbC) since, to this day, we still have not seen any improvements in the short or long-term mortality rate.2 However, its use may be an option on a per-patient basis in to achieve hemodynamical stability, guarantee the proper perfusion of vital organs or as bridging therapy for the recovery of myocardial function.
Given the presence of complete atrioventricular block and severe hypotension, in our case we considered implanting the Impella CP device (AbioMed, Danvers, Massachusetts, United States), which was the device that provided the highest utility since it does not require arterial pulse or an electrocardiographic registry to operate; however, performing a transthoracic echocardiography prior to the implantation of the device could have been decisive for diagnostic purposes and to re-think what was the best therapeutic strategy to use since, probably, the clinical situation of the patient was not only due to the STEMI, but also to the obstructive effect of the mass and the systemic embolization that may have exacerbated after the implantation of the device.
With this case we learned that performing a transthoracic echocardiography in a patient with cardiogenic shock before approaching other therapeutic attitudes is essential. Also, that the delay involved should not care at all given the benefits derived from finding possible contraindications as it happened with our case.
REFERENCES
1. Panos A, Kalangos A, Sztajzel J. Left atrial myxoma presenting with myocardial infarction. Case report and review of the literature. Int J Cardiol. 1997;62:73-75.
2. Ouweneel DM, Eriksen E, Sjauw KD, et al. Percutaneous mechanical circulatory support versus intra-aortic balloon pump in cardiogenic shock after acute myocardial infarction. J Am Coll Cardiol. 2017;69:278-287.
Corresponding author: Servicio de Cardiología, Àrea de Malalties del Cor, Hospital Universitario de Bellvitge, Feixa Llarga s/n, 08907 L’Hospitalet de Llobregat, Barcelona, Spain.
E-mail addresses: maserrit@msn.com; mgracida@bellvitgehospital.cat (M. Gracida).