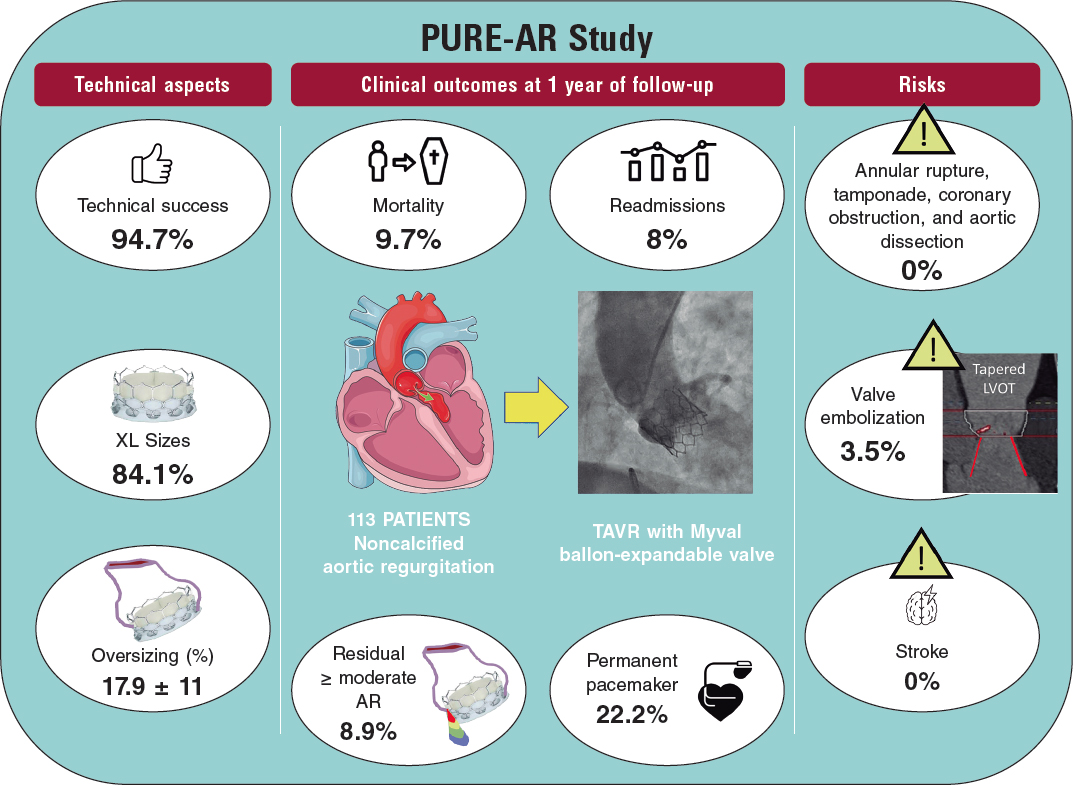
ABSTRACT
Introduction and objectives: Complex calcified lesions can affect stent expansion and lead to stent failure and adverse outcomes. Intracoronary lithotripsy (ICL) has emerged as a new tool that enables calcium modification. The Disrupt CAD II clinical trial has recently evaluated the safety and feasibility of ICL in patients with stable coronary disease and calcified coronary lesions. Although its use has increased rapidly, the experience already reported with this new device is limited. We report the results in real-life complex patients with heavy coronary calcification.
Methods: From October 2018 to March 2019, 25 patients (37 calcified lesions) were treated in 2 Spanish centers, which accounted for 2.7% of the patients treated with percutaneous coronary intervention.
Results: The device and clinical success rates were 84% and 95%, respectively. No procedure-related complications were seen. The crossing rate of the ICL balloon was 100% and balloon rupture during inflation occurred in 8%. The ICL was performed in a subset of highly complex lesions like left main coronary artery lesions and chronic total coronary occlusions. Compared to the Disrupt CAD II trial, our patients were younger but their clinical scenario was worse with a higher prevalence of diabetes (68%), renal failure (22%), and up to 76% suffered from acute coronary syndrome. The ICL failed to reach proper expansion in 3 out of 4 cases of stent underexpansion. The procedure was performed safely, and clinical and device success were high with no in-hospital mortality. One patient died of non-cardiac causes at the 30-day follow-up.
Conclusions: The ICL-assisted percutaneous coronary intervention was performed safely and effectively in a real-life cohort of patients with calcified and highly complex lesions.
Keywords: Lithotripsy. Calcium. Shockwave.
RESUMEN
Introducción y objetivos: Las lesiones coronarias calcificadas pueden impedir una correcta expansión del stent que en ocasiones conduce a eventos adversos. La litotricia intracoronaria es una nueva herramienta de modificación de la placa, cuyas seguridad y viabilidad en pacientes con enfermedad coronaria estable han sido evaluadas en el ensayo Disrupt CAD II. Aunque su uso ha aumentado rápidamente, hasta el momento solo se han comunicado casos aislados en escenarios concretos. Se presentan los resultados en pacientes clínicamente complejos de la vida real con calcificación coronaria grave.
Métodos: Entre octubre de 2018 y marzo de 2019 se trató a 25 pacientes (37 lesiones) en 2 centros españoles, lo que representa el 2,7% de los pacientes tratados con intervención coronaria percutánea.
Resultados: Las tasas de éxito clínico y del dispositivo fueron del 84 y el 95%, y no se observaron complicaciones relacionadas con el procedimiento. En todos los casos se consiguió cruzar la lesión con el balón de litotricia intracoronaria, si bien en el 8% de los casos se rompió el balón durante el inflado. Se trataron con éxito lesiones complejas, como oclusiones coronarias totales y estenosis del tronco común. En comparación con el estudio Disrupt CAD II, nuestros pacientes eran más jóvenes, pero tenían peor escenario clínico, con mayor prevalencia de diabetes (68%) e insuficiencia renal (22%), y hasta el 76% se presentó como síndrome coronario agudo. En 3 de 4 pacientes con infraexpansión de stent tratados con litotricia intracoronaria no se consiguió una expansión adecuada tras el procedimiento. No hubo complicaciones ni mortalidad hospitalaria. Un paciente falleció por causa no cardiaca a los 30 días de seguimiento.
Conclusiones: La litotricia intracoronaria se ha demostrado efectiva y segura en una cohorte de pacientes complejos de la vida real con lesiones calcificadas.
Palabras clave: Litotricia intracoronaria. Calcio. Ondas de choque.
Abreviaturas CTO: chronic total coronary occlusion. ICL: intracoronary lithotripsy. OCT: optical coherence tomography. PCI: percutaneous coronary intervention.
INTRODUCTION
Percutaneous coronary intervention (PCI) in calcified coronary lesions is often challenging and may be associated with suboptimal stent expansion and apposition both related to stent failure due to stent thrombosis and in-stent restenosis.1-3 The balloon angioplasty used in calcified lesions increases the risk of dissection of non-calcified segments usually without significant modification of calcified plaques and often without a proper luminal gain.4 The management of this subset of lesions is complex and often requires complex techniques such as rotational atherectomy or excimer laser coronary atherectomy.
Intracoronary lithotripsy (ICL) (Shockwave Medical, Freemont, CA, United States) has emerged as a new tool to modify calcium by applying a diffuse acoustic pulse through a balloon inflated at 4 to 6 atmospheres without damage to endovascular soft tissues. The multicenter, prospective, single-arm Disrupt CAD II clinical trial5 has recently evaluated the safety and feasibility of the ICL system prior to stent implantation in 120 patients with coronary artery disease and calcified coronary lesions. This study showed that the ICL appeared feasible with favorable initial success and complication rates in selected patients.5 Although its use has grown rapidly among interventional cardiologists and there are many case reports on the medical literature available to us, the experience already reported on this new device is quite limited. We present the initial results of lithotripsy-assisted PCIs in a real-life cohort of high-risk patients with complex, calcified lesions.
METHODS
Patient population and data collection
Two-center, prospective, observational registry including all consecutive PCI cases that required ICL prior to stent implantation to the operator’s discretion from October 2018 to March 2019. The baseline characteristics and procedural and in-hospital outcomes were prospectively recorded.
Intracoronary lithotripsy procedure
The ICL system is a portable and rechargeable generator connected to the ICL catheter. The catheter consists of a rapid exchange semi-compliant 12-mm balloon with 2 radiopaque emitters mounted inside available in 2.5, 3.0, 3.5, and 4 mm diameters. The catheter is compatible with a 6-Fr guiding catheter with a crossing profile range of 0.042 in and it is placed across the calcified lesion through a 0.014 in guidewire. Once in position, the balloon is inflated at 4 atmospheres to make intimate contact with the vessel wall and facilitate an efficient the transfer of energy. An electrical discharge from the emitters vaporizes the fluid inside the balloon generating sonic pressure waves that create a localized field effect. The ICL catheter is connected to a generator preprogrammed to deliver 10 pulses at a rate of 1 pulse per second. Each catheter can administer a maximum of 80 pulses. The sonic pulses through the soft vascular tissue cause selective microfractures at the intimal and medial calcium level of the vessel wall. After the pulse emission and the corresponding modification of calcium, the balloon is inflated up to 6 atmospheres to maximize luminal gain.
Definitions and outcomes
The use of the ICL catheter was based on the presence of a significant and severely calcified lesion (70% stenosis in an epicardial coronary vessel) on the angiography or intravascular imaging.
Coronary calcified lesions were defined by: a) the presence of radiopacities prior to contrast injection often involving both sides of the arterial wall; b) the presence of ≥ 270 degrees of calcium on at least one single cross-section on the intravascular ultrasound or optical coherence tomography (OCT); or c) subsets of calcified lesions with previous failed revascularization attempts.
According to the Disrupt CAD II trial criteria,5 lithotripsy delivery was considered successful when it facilitated stent delivery with < 50% residual stenosis and without any serious angiographic complications like severe dissection, perforation, slow flow or persistent no-reflow. In addition, clinical success was defined as residual stenosis < 50% after stenting without any evidence of in-hospital adverse events. We also assessed procedural complications such as PCI-related myocardial infarction (type 4a myocardial infarction, defined according to the fourth universal definition of myocardial infarction),6 and in-hospital and 30-day outcomes.
Statistical analysis
Categorical variables were expressed as number (percentage) and continuous variables as mean ± standard deviation or median according to their distribution. We analyzed all data using the STATA statistical package version 15.0 (StataCorp LP, College Station, Texas, United States).
RESULTS
Patients
Between October 2018 and March 2019, 25 patients with 37 calcified lesions were treated, which amounted to 2.7% of the patients treated with PCIs in both centers. The baseline characteristics of the patients are shown on table 1. Mean age was 71 ± 9 years and 68% of the patients were males. The traditional cardiovascular risk factors were common and the vast majority of patients had undergone a previous revascularization (PCI or coronary artery bypass graft). The indication for PCI was acute coronary syndrome in most cases (76%), all of them non–ST-elevation myocardial infarctions.
Table 1. Baseline characteristics (per patient)
| Clinical characteristics | N = 25 |
|---|---|
| Age, years | 71 ± 9 |
| Male sex | 17 (68) |
| Diabetes | 17 (68) |
| Renal failure | 7 (28) |
| Peripheral vascular disease | 8 (32) |
| Previous PCI | 14 (56) |
| Previous CABG | 3 (12) |
| LVEF | 49 ± 17 |
| ACS on admission | 19 (76) |
ACS, acute coronary syndrome; CABG, coronary artery bypass graft; LVEF, left ventricular ejection fraction; PCI, percutaneous coronary intervention. Data are expressed as no. (%) or mean ± standard deviation. | |
Procedural characteristics
Procedural characteristics are shown on table 2. The mean SYNTAX score (Synergy between Percutaneous Coronary Intervention with Taxus and Cardiac Surgery) was 19.3 ± 2, and the left anterior descending artery was the most-treated vessel. A non-negligible proportion of complex coronary artery lesions like chronic total coronary occlusions (CTO), bifurcation and ostial lesions, and stent underexpansion were also treated.
Table 2. Procedural characteristics
| Lesion characteristics | N = 37 |
|---|---|
| Protected LMCA | 1 (3) |
| Unprotected LMCA | 4 (11) |
| LAD | 17 (46) |
| LCx | 3 (8) |
| RCA | 12 (32) |
| Syntax Score | 19.3 ± 2 |
| Stent underexpansion treatment | 4 (11) |
| Ostial lesions | 13 (35) |
| Bifurcation lesion | 13 (35) |
| CTO | 3 (8) |
| Lesion severity by QCA | N = 37 |
| Pre-PCI diameter stenosis | 81.6 ± 2.5 |
| Post-PCI diameter stenosis | 15.9 ± 3.4 |
| Pre-PCI area stenosis | 84.6 ± 3.8 |
| Post-PCI area stenosis | 21.6 ± 3.5 |
| Total lesion length, mm | 20.7 ± 3 |
| Mean luminal diameter, mm | 0.77 ± 0.1 |
| Procedural characteristics | N = 25 |
| Radial access | 15 (60) |
| Mechanical support Impella device | 5 (20) |
| Fluoroscopy time, min | 31.5 ± 4.8 |
| Contrast, mL | 212 ± 28 |
| Number of vessels treated (per patient) | 1.3 ± 0.5 |
| Number of lesions treated (per patient) | 1.7 ± 0.8 |
| PCI characteristics | N = 37 |
| Pre-ICL balloon pre-dilatation | 23 (62) |
| Pre-ICL rotational atherectomy | 10 (27) |
| Pre-ICL cutting balloon | 2 (5) |
| ICL number of pulses | 46 ± 19 |
| ICL balloon rupture | 3 (8%) |
| Number of stents implanted | 1.2 ± 0.6 |
| Stent diameter, mm | 3.3 ± 1 |
| Stent length, mm | 23.1 ± 10 |
| Angiographical success | 35 (95) |
CTO, chronic total coronary occlusion; ICL, intracoronary lithotripsy; LAD, left anterior descending coronary artery; LCx, left circumflex artery; LMCA, left main coronary artery; PCI, percutaneous coronary intervention; QCA, quantitative coronary angiography; RCA, right coronary artery. Data are expressed as no. (%) or mean ± standard deviation. The ICL was used after a failed attempt of balloon pre-dilatation in 62% of the lesions, of which 10% showed balloon rupture. Ten lesions (27%) had previously been treated with rotational atherectomy. Only one lithotripsy catheter per lesion was required, and the mean number of pulses was 46 ± 19. The crossing rate of the lithotripsy balloon was 100% in pre-dilated and non-pre-dilated lesions, and the ICL balloon rupture occurred in 3 cases (8%) with no associated complications. An OCT prior to the ICL therapy was performed in a small percentage of cases (10%) to the operator’s discretion. All stents implanted were drug-eluting stents and successful angiographic result according to the definition was achieved in 95% of cases. | |
Intracoronary lithotripsy in complex lesions
A subset of complex lesions was also treated with lithotripsy balloon (table 2). Success rate was 100% for left main coronary artery revascularizations, 100% for CTOs, and 86% for bifurcations.
Left main coronary artery lesions
Five patients with left main coronary artery lesions were treated with the ICL balloon. Four were unprotected lesions and were treated under hemodynamic support using the Impella device (all of them showed a severely depressed ejection fraction and/or a right coronary artery chronic total occlusion). Device and clinical success were achieved in all cases.
Bifurcation lesions
Seven lesions treated involved bifurcations, 4 were treated using a provisional stenting technique, and 3 cases were treated using the 2-stent technique (V stenting).
Chronic total coronary occlusions
The CTOs of 3 patients were treated using the ICL with complete success in all of them. The first patient had a severely calcified aorto-ostial lesion in the right coronary artery, a Japanese chronic total coronary occlusion score (Japanese Multicenter CTO Registry) of 2 (presence of calcification ≥ 20 mm in length). After unsuccessful pre-dilation using 2 balloons (1 of them ruptured) the ICL was performed and good balloon expansion was achieved without need for post-dilatation prior to the stenting. The second successfully treated CTO case involved the mid portion of the left anterior descending coronary artery (bifurcation according to the Medina classification 1,1,1), Japanese CTO score of 2 (calcification ≥ 20 mm in length) that had been treated with rotational atherectomy prior to the ICL. The third case was the CTO of a distal right coronary artery involving bifurcation (according to the Medina classification 1,0,0) and a Japanese CTO score of 3 (blunt-tip entry, calcification ≥ 20 mm in length). The artery was dilated using 5 balloons, some of which ruptured before performing the ICL. Lesion expansion was completed with a cutting balloon after the ICL, which allwed proper stent implantation.
Stent underexpansion
Four cases of stent underexpansion were treated, but results after the ICL were only successful in one case. There was 1 case that needed additional very high-pressure balloon dilatation (up to 40 atmospheres) for proper expansion, and 2 cases that remained unexpanded even after very high-pressure balloon dilatation (up to 40 atmospheres), and in-stent rotational atherectomy with 1.75 and 2.00 mm burrs.
In-hospital and 30-day outcomes
The procedure was performed safely in all cases. Both the clinical and device success were high with no in-hospital mortality. One patient died of non-cardiac causes at the 30-day follow-up (sepsis due to spontaneous bacterial peritonitis in the presence of hepatic cirrhosis). Procedural, in-hospital, and 30-day outcomes are shown on table 3.
Table 3. In-hospital and 30-day outcomes
| Clinical outcomes | N = 25 |
|---|---|
| Procedural complications | |
| Dissection | 0 (0) |
| Perforation | 0 (0) |
| No-reflow | 0 (0) |
| Type 4a acute myocardial infarction | 3 (12) |
| In-hospital mortality | 0 (0) |
| 30-d myocardial infarction | 0 (0) |
| 30-d target-vessel revascularization | 0 (0) |
| 30-d stent thrombosis | 0 (0) |
| 30-d mortality | 0 (0) |
| Cardiac death | 0 (0) |
| Non-cardiac death | 1 (4) |
Data are expressed as no. (%) or mean ± standard deviation. | |
DISCUSSION
We present our initial experience with ICL in a cohort of real-life clinically complex patients with heavy coronary artery calcification and showed that the ICL is feasible with favorable initial outcomes and low complication rates.
Debulking techniques like rotational atherectomy, orbital atherectomy or excimer laser coronary atherectomy are commonly used to treat calcified coronary lesions. Back in 2018, in Spain up to 1517 patients were treated with rotational atherectomy and 88 with excimer laser coronary atherectomy.7 Recently, the ICL has emerged as an attractive option for the management of patients with severely calcified coronary lesions. Nevertheless, the experience reported on this new technique is still limited. The recently published single-arm Disrupt CAD II clinical trial confirmed the safety and performance of ICL to treat calcified coronary lesions.5 However, the clinical characteristics of the patients enrolled in this trial show a relatively low-risk population. Complex calcified coronary lesions are a common thing and they amount to 25% to 30% of all PCIs performed.3 Among our population, 2.7% of patients were considered eligible to receive ICL therapy, indicative of a highly demanding indication criterion. Compared to the Disrupt CAD II clinical trial5 our patients were younger but had a worse clinical scenario with a higher prevalence of diabetes (68% vs 32%) and renal failure (22% vs 9%), and up to 76% had suffered an acute coronary syndrome (none in the Disrupt CAD II trial). Another recent report described the initial experience with ICL in a cohort of 26 patients with calcified coronary lesions with findings for the clinical characteristics and results similar to the Disrupt CAD II.8
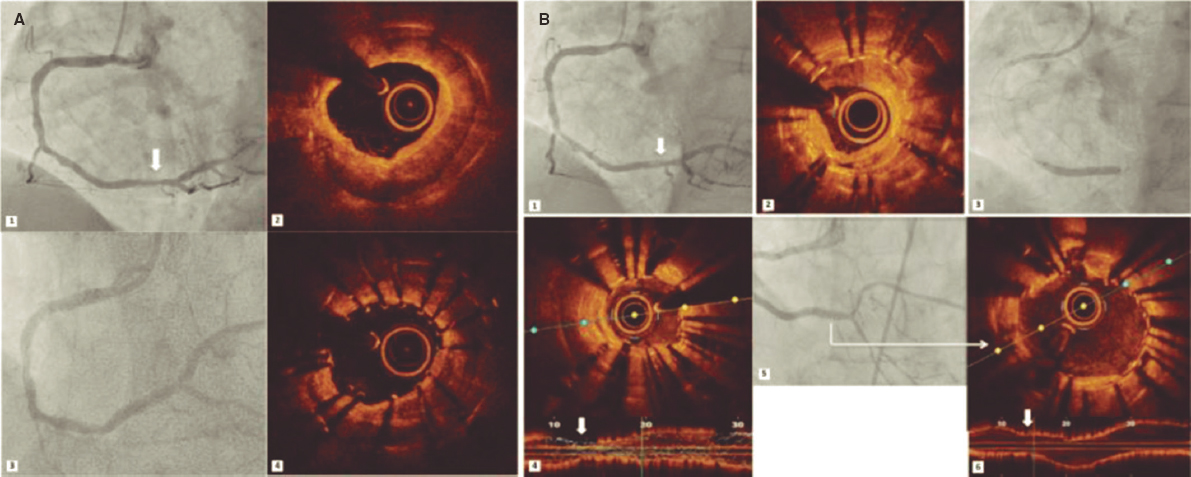
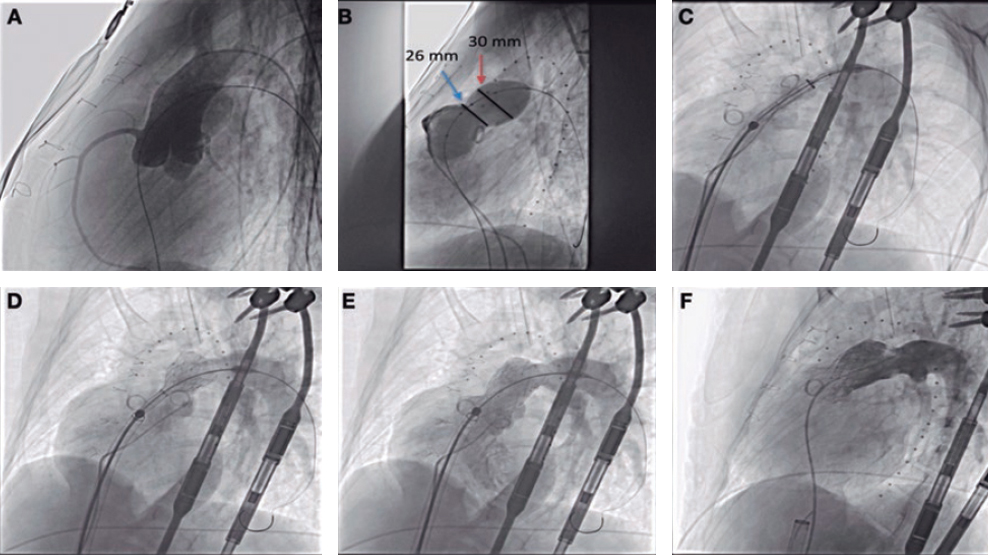
Regarding the procedure, it should be noted that the crossing rate for the ICL balloon was 100% despite a high percentage of plaque preparation was required (62% balloon pre-dilatation, 27% rotational atherectomy). Recently, the combination of rotational atherectomy and ICL has been described as RotaTripsy, suggestive that these 2 calcium debulking techniques may be complementary, since rotational atherectomy facilitates the ICL balloon crossing, and the latter facilitates proper expansion in the presence of circumferential deep calcium plaques.9 The device success rate was 84% (100% in the Disrupt CAD II linical trial) and the clinical success rate was 95% (94% in the Disrupt CAD II trial). And most important of all, no major procedural complications were seen, which is consistent with the Disrupt CAD II trial results. The rupture of the ICL balloon during inflation occurred in 3 cases (12%) without associated complications, yet the rupture of the balloon has been described in a case report resulting in a type C coronary dissection; the interventional cardiologist needs to be aware of this lithotripsy-related potential complication.10 Intravascular imaging were performed in few cases probably because the operator thought it would be difficult to cross an especially severe and calcified lesion with the OCT or IVUS catheter. Consistent with the results of the Disrupt CAD I and II clinical trials and OCT substudy,5,11 it was confirmed that the modification of calcium and the presence of fractures lead to an acute area gain and favorable stent expansion in the lesions assessed through OCT in our series. Figure 1 shows the coronary angiography and OCT of one complex patient treated with ICL; the red arrows seen on figure 1D,E indicate calcium fractures after the ICL.
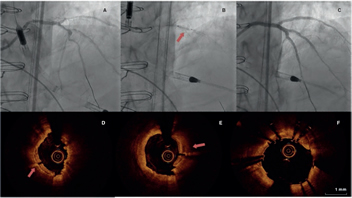
Figure 1. Intracoronary lithotripsy, angiography, and optical coherence tomography. Patient with severe coronary artery heart disease with severely depressed left ventricular ejection fraction previously treated with coronary artery bypass graft (venous graft-left anterior descending coronary artery, currently occluded). Treatment of left main coronary artery, left anterior descending artery, and diagonal branches. A: pre-intracoronary lithotripsy angiography. B: Impella-assisted PCI of left anterior descending coronary artery. Arrow indicates inflated lithotripsy balloon. C: successful final angiographic result after stenting. D, E and F: optical coherence tomography cross-sectional images of a post-lithotripsy calcified lesion. Red arrows indicate calcium microfractures after intracoronary lithotripsy.
We used the ICL in a subset of highly complex lesions like left main coronary artery stenosis, CTO, stent underexpansion, and bifurcation lesions.
Five complex patients with calcified left main coronary artery and severe stenosis were treated with ICL; mechanical support with the Impella device was needed in 4 patients due to a depressed left ventricular ejection fraction. Recently, a case report with 2 patients that were successfully treated with ICL in a left main coronary artery stenosis has also been published.12 The ICL seems like a safe treatment option to treat calcified left main coronary artery stenoses even in technically complex cases that require hemodynamic support, a clinical scenario where the use of rotational atherectomy or excimer laser coronary atherectomy is rare.
Three patients with CTO lesions were successfully treated with ICL. Treatment of CTO with ICL has been previously described in 2 case reports. The first one was a patient with a CTO in the proximal right coronary artery. In this case, the ICL allowed the reverse controlled antegrade/retrograde tracking at the location of heavy calcification at the site of the chronic occlusion.13 The second case was a patient with a proximal right coronary artery CTO due to heavily calcified in-stent restenosis. The ICL achieved good lesion expansion prior to stent implantation.14 If performed properly, the ICL can be an alternative to other debulking techniques in heavily calcified CTO lesions to guarantee proper lesion expansion.
Several case reports of stent underexpansion due to heavily calcified lesions successfully treated with ICL have been reported recently.15,16 Surprisingly, in our series the ICL failed to achieve proper stent expansion in 3 out of the 4 cases attempted. The management of stent underexpansion using ICL should be performed with extra caution because sound waves can damage the metallic structure of the stent.
The management of calcified bifurcation lesions is often complex due to the high risk of side branch occlusion when applying debulking techniques such as rotational atherectomy or excimer laser coronary atherectomy because the treatment cannot be performed using a guidewire for side branch protection purposes.17,18 The ICL allows us to treat the main branch bifurcation with a guidewire in the side branch to guarantee quick access in case of flow impairment just like conventional procedures do.
Compared to atherectomy or specialty balloons, the ICL is said to offer several potential advantages5 and requires no specific training as the device is delivered similar to the standard catheter-based PCI. ICL therapy is balloon based, and, therefore, the risk of atheromatous embolization may be lower compared to free debulking devices; according to the Disrupt CAD I (19) or Disrupt CAD II trial5 results, none of the patients from our series experienced no-reflow events and the rate of periprocedural myocardial infarction was relatively low. Whereas standard and specialty balloons are inflated at high atmospheric pressure to modify calcium, the ICL is typically performed at low atmospheric pressure balloon inflation, thus minimizing mechanical vascular trauma. Lastly, side-branch protection using a guidewire may be easily performed using ICL, without running the risk of wire entrapment or severing associated with rotational or orbital atherectomy. However, there is no evidence regarding stent restenosis of lesions treated with ICL therapy. New studies like the Disrupt CAD III trial that has just begun and will be recruiting up to 400 patients with a 2-year follow-up are needed to determine long-term outcomes.
Limitations
This 2-center experience using the ICL balloon has the limitations inherent to an observational study with a small sample size, which limits drawing conclusions especially in subgroups of high-risk lesions treated with ICL. However, in our opinion, it may contribute by adding more evidence supporting the use of ICL. This study did not have a comparison group either among existing plaque-modifying techniques.
CONCLUSIONS
In our own experience, the ICL-enhanced PCI was performed safely and effectively in a real-life cohort of complex patients with severely calcified and highly complex lesions.
CONFLICTS OF INTEREST
The authors of the manuscript declared no conflicts of interest.
WHAT IS KNOWN ABOUT THE TOPIC?
Calcified lesions continue to be a challenge for interventional cardiologists since poor plaque preparation prevents proper stent expansion, which leads to a higher rate of periprocedural complications and long-term adverse events.
The ICL balloon is a new plaque modification tool whose safety and efficacy in patients with stable coronary heart disease has recently been evaluated in a cohort of 120 patients in the Disrupt CAD II clinical trial.
The use of the ICL balloon has grown rapidly in the cardiac catheterization laboratories. However, to this day extensive series in real-life patients have not been reported yet.
WHAT DOES THIS STUDY ADD?
We present the results of the ICL balloon in real-life patients referred to undergo coronary angioplasty.
Although the size of the sample in our series was not big enough to draw any conclusions, the patients included were clinically complex and a high percentage of acute coronary syndromes and technically complex interventions (left main coronary artery lesions, bifurcation lesions, CTOs and stent underexpansion) was reported.
The procedure was performed safely and successfully in a large percentage of cases (95%). In-hospital mortality was zero and only one patient died at the 30-day follow-up (due to non-cardiac reasons).
REFERENCES
1. Bourantas CV, Zhang YJ, Garg S, et al. Prognostic implications of coronary calcification in patients with obstructive coronary artery disease treated by percutaneous coronary intervention:a patient-level pooled analysis of 7 contemporary stent trials. Heart. 2014;100:1158-1164.
2. Fitzgerald PJ, Ports TA, Yock PG. Contribution of localized calcium deposits to dissection after angioplasty. An observational study using intravascular ultrasound. Circulation. 1992;86:64-70.
3. Khattab AA, Otto A, Hochadel M, Toelg R, Geist V, Richardt G. Drug-eluting stents versus bare metal stents following rotational atherectomy for heavily calcified coronary lesions:late angiographic and clinical follow-up results. J Interv Cardiol. 2007;20:100-106.
4. Mehanna E, Bezerra HG, Prabhu D, et al. Volumetric characterization of human coronary calcification by frequency-domain optical coherence tomography. Circ J. 2013;77:2334-2340.
5. Ali ZA, Nef H, Escaned J, et al. Safety and effectiveness of coronary intravascular lithotripsy for treatment of severely calcified estenoses. The Disrupt CAD II Study. Circ Cardiovasc Interv. 2019;12:e008434.
6. Thygesen K, Alpert JS, Jaffe AF, et al. ESC Scientific Document Group;Fourth universal definition of myocardial infarction (2018). Eur Heart J. 2019;40:237-269.
7. Cid-Alvarez AB, Rodriguez-Leor O, Moreno R, Pérez de Prado A. Registro Español de Hemodinámica y Cardiología Intervencionista. XXVIII Informe Oficial de la Sección de Hemodinámica y Cardiología Intervencionista de la Sociedad Española de Cardiología (1990-2018). Rev Esp Cardiol. 2019. https://doi.org/10.1016/j.recesp.2019.07.023.
8. Wong B, El-Jack S, Newcombe R, Glenie T, Armstrong G, Khan A. Shockwave Intravascular Lithotripsy for Calcified Coronary Lesions:First Real-World Experience. J Invasive Cardiol. 2019;31:46-48.
9. Jurado-Román A, Gonzálvez A, Galeote G, Jiménez-Valero S, Moreno R. RotaTripsy:Combination of Rotational Atherectomy and Intravascular Lithotripsy for the Treatment of Severely Calcified Lesions. JACC Cardiovasc Interv. 2019;12:e127-e129.
10. López-Lluva MT, Jurado-Román A, Sánchez-Pérez I, Abellán-Huerta J, Lozano Ruíz-Poveda F. Shockwave:Useful But Potentially Dangerous. JACC Cardiovasc Interv. 2019;12:500-501.
11. Ali ZA, Brinton TJ, Hill JM, et al. Optical coherence tomography characterization of coronary lithoplasty for treatment of calcified lesions:first description. JACC Cardiovasc Imaging. 2017;10:897-906
12. Salazar CH, Travieso A, Gonzalo N, Escaned J. Intracoronary lithotripsy in percutaneous treatment of calcified left main coronary stenoses. JACC:Case Rep. 2019;1:46-49.
13. Yeoh J, Hill J, Spratt JC. Intravascular lithotripsy assisted chronic total occlusion revascularization with reverse controlled antegrade retrograde tracking. Catheter Cardiovasc Interv. 2019;93:1295-1297.
14. Azzalini L, Bellini B, Montorfano, Carlino M. Intravascular lithotripsy in chronic total occlusion percutaneous coronary intervention. EuroIntervention. 2019. https://doi.org/10.4244/EIJ-D-19-00175.
15. Ali ZA, McEntegart M, Hill JM, Spratt JC. Intravascular lithotripsy for treatment of stent under expansion secondary to severe coronary calcification. Eur Heart J. 2018. https://doi.org/10.1093/eurheartj/ehy747.
16. Watkins S, Good R, Hill J, Brinton TJ, Oldroyd KG. Intravascular lithotripsy to treat a severely under-expanded coronary stent. EuroIntervention. 2019;15:124-125.
17. Rawlins J, Din JN, Talwar S, O'Kane P. Coronary intervention with the Excimer Laser:review of the technology and outcome data. Interv Cardiol. 2016;11:27-32.
18. Nageh T, Kulkarni NM, Thomas MR. High-speed rotational atherectomy in the treatment of bifurcation-type coronary lesions. Cardiology. 2001;95:198-205.
19. Brinton TJ, Ali ZA, Hill JM, et al. Feasibility of shockwave coronary intravascular lithotripsy for the treatment of calcified coronary stenosis. Circulation. 2019;139:834-836.
Corresponding author: Departamento de Cardiología Intervencionista, Institut del Cor (ICOR), Hospital Germans Trias i Pujol, Ctra. de Canyet s/n, 08916 Badalona, Barcelona, Spain.
E-mail address: oriolrodriguez@gmail.com (O. Rodríguez-Leor).